Description
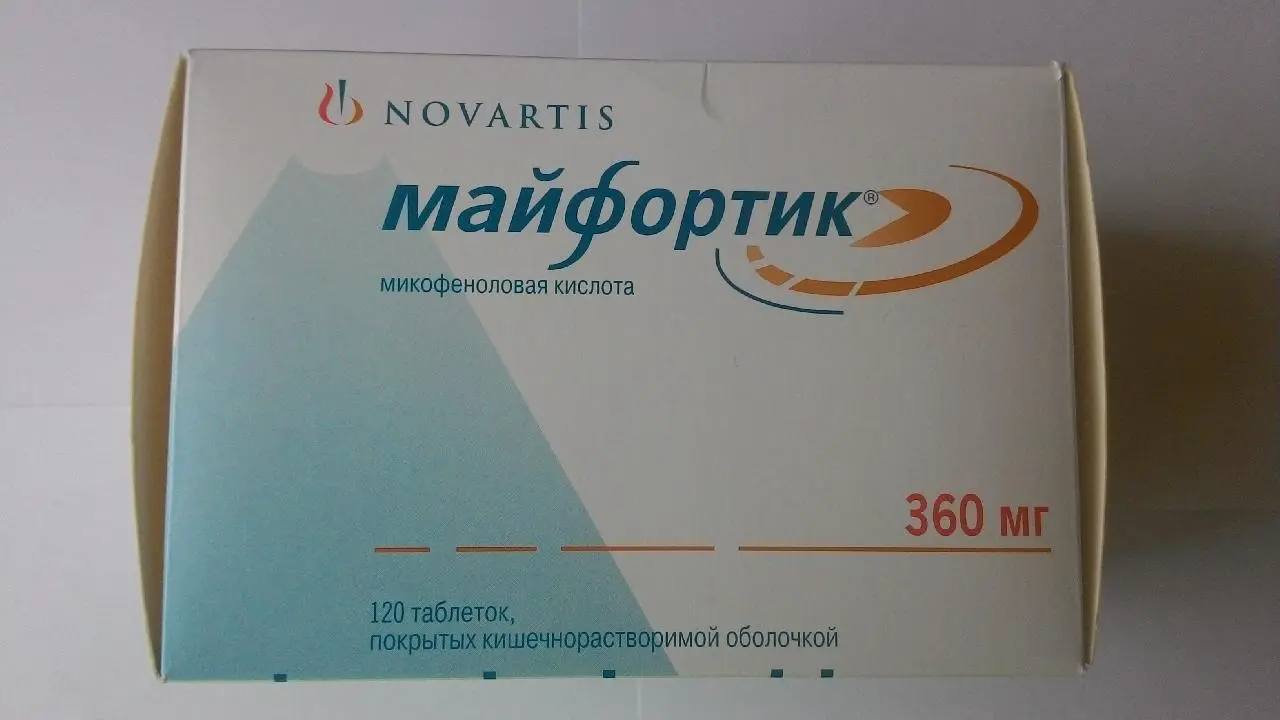
Mifortik (mycophenolate sodium) Enteric Tablets 180 mg.
Mifortik is a medication that contains mycophenolate sodium as the active ingredient. It is available in the form of enteric-coated tablets with a strength of 180 mg. Each pack contains 120 tablets, providing a convenient supply for patients requiring this medication.
What is Mycophenolate Sodium?
Mycophenolate sodium is a medication that belongs to a class of drugs known as immunosuppressants. It is commonly used in transplant medicine to prevent the rejection of transplanted organs such as kidney, heart, or liver. Mycophenolate sodium works by suppressing the immune system to reduce the risk of the body attacking the newly transplanted organ.
Indications
Mifortik enteric tablets are indicated for the prevention of organ rejection in patients receiving kidney, heart, or liver transplants. It is usually used in combination with other immunosuppressive medications as part of a comprehensive transplant regimen.
How to Take Mifortik Tablets
It is important to follow your healthcare provider’s instructions when taking Mifortik tablets. The usual dose of mycophenolate sodium is determined based on your medical condition, response to treatment, and other medications you may be taking. The tablets should be taken by mouth with a full glass of water, usually once or twice daily.
It is recommended to take Mifortik tablets on an empty stomach, at least 1 hour before or 2 hours after a meal, to maximize the absorption of the medication. Do not crush, chew, or break the tablets, as they are enteric-coated to protect the stomach lining from irritation.
Possible Side Effects
Like all medications, Mifortik tablets may cause side effects in some patients. Common side effects of mycophenolate sodium may include:
- Nausea
- Vomiting
- Diarrhea
- Headache
- Stomach pain
If you experience severe or persistent side effects while taking Mifortik tablets, contact your healthcare provider for further evaluation and guidance.
Precautions and Warnings
Before taking Mifortik tablets, inform your healthcare provider about any medical conditions you have, especially if you have a history of infections, liver disease, or digestive problems. It is important to disclose all medications you are currently taking, including over-the-counter drugs, herbal supplements, and vitamins, to avoid potential drug interactions.
Do not use Mifortik tablets if you are pregnant, planning to become pregnant, or breastfeeding, as mycophenolate sodium may harm the unborn baby or pass into breast milk. Use effective contraception during treatment with this medication to prevent pregnancy.
Storage and Handling
Store Mifortik tablets at room temperature away from moisture and heat. Keep the medication in its original packaging to protect it from light and humidity. Do not use Mifortik tablets beyond the expiration date printed on the packaging, and dispose of any unused or expired medication properly according to local regulations.
Conclusion
Mifortik (mycophenolate sodium) enteric tablets 180 mg. are a valuable medication used in transplant medicine to prevent organ rejection in patients undergoing kidney, heart, or liver transplants. By following your healthcare provider’s instructions and taking the medication as directed, you can maximize the benefits of Mifortik while minimizing the risk of potential side effects. If you have any questions or concerns about Mifortik tablets, do not hesitate to consult your healthcare provider for personalized guidance and support.